Every year, millions of prescription drugs move through a complex network of manufacturers, distributors, wholesalers, and pharmacies before they land in your medicine cabinet. But what if one of those pills wasn’t made in a regulated lab? What if it was fake - laced with the wrong ingredients, or worse, nothing at all? The DSCSA track-and-trace system was built to stop that before it ever happens.
What Is the DSCSA, and Why Does It Matter?
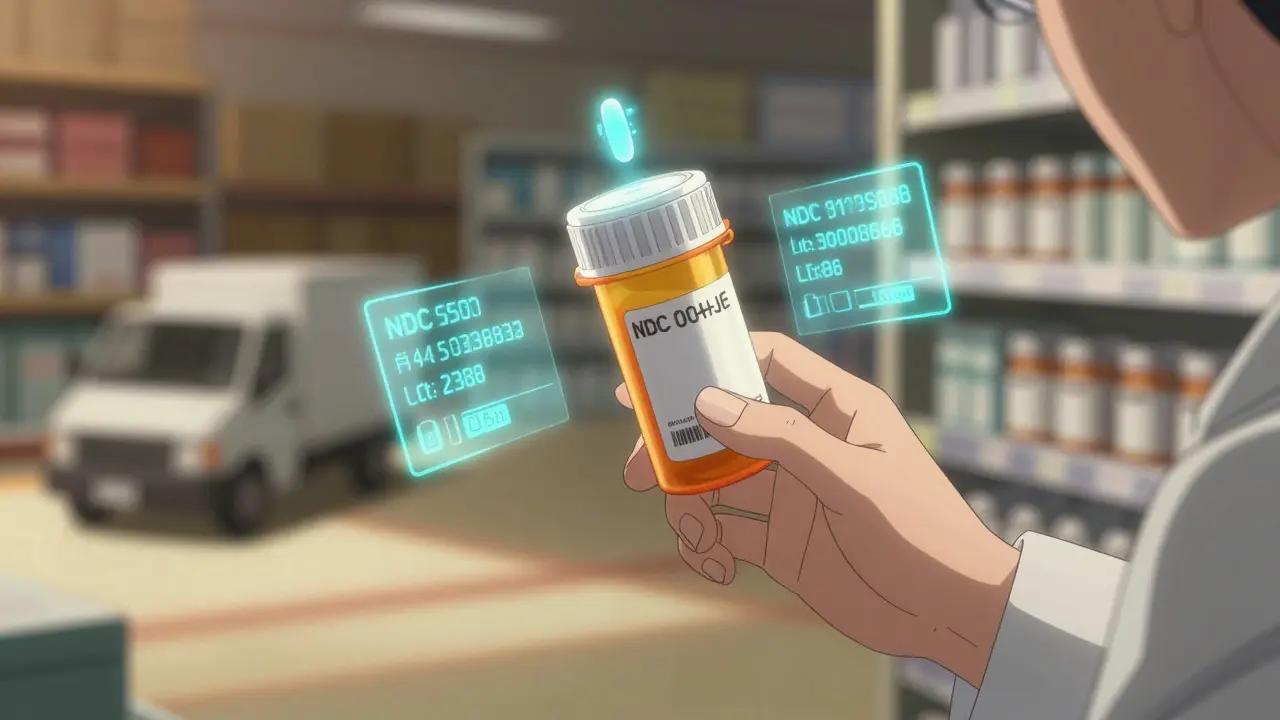
The Drug Supply Chain Security Act (DSCSA) is a federal law passed in 2013 to protect Americans from counterfeit, stolen, or contaminated drugs. Before DSCSA, there was no nationwide system to track prescription medications from factory to pharmacy. Each state had its own rules - or none at all. That patchwork made it easy for fake drugs to slip through. A single counterfeit batch could end up in dozens of states before anyone noticed. DSCSA changed that. It created one clear, uniform standard across the entire U.S. pharmaceutical supply chain. The goal? To make every prescription drug package uniquely identifiable. That means every bottle, box, or vial gets a serial number, lot number, expiration date, and National Drug Code (NDC) - all printed in both human-readable text and machine-scannable barcode format. By November 27, 2024, every pharmacy, wholesaler, and manufacturer had to be fully connected to an electronic system that can verify each package’s authenticity in seconds. This isn’t just paperwork. It’s a lifeline. The FDA estimates that DSCSA has already cut counterfeit drug incidents by 95% since its rollout began. That’s not a guess - it’s based on real data from verified product checks and reduced recalls.How the DSCSA Track-and-Trace System Works
Think of DSCSA like a digital passport for every pill. Here’s how it works step by step:- Manufacturer: When a drug is packaged, a unique 20-character serial number is printed on it using GS1 standards. This number is tied to the product’s NDC, lot, and expiration date.
- Wholesaler: When the drug moves from the manufacturer to a distributor, the wholesaler scans the barcode and records the transaction electronically. They send three pieces of data: Transaction Information (TI), Transaction History (TH), and Transaction Statement (TS). Together, these prove the drug’s path is legal and untampered.
- Pharmacy: When the drug arrives at your local pharmacy, the pharmacist scans the barcode. Their system checks the serial number against the manufacturer’s database. If the number doesn’t match - or if the product was never registered - the system flags it immediately.
Who’s Required to Comply?
DSCSA doesn’t just target big players. It covers everyone in the chain:- Manufacturers: Must serialize every package and provide electronic transaction data.
- Repackagers: If a company repackages bulk drugs into smaller doses (like for nursing homes), they must also serialize and track.
- Wholesalers: Must verify the legitimacy of every product they receive and forward transaction data to the next link.
- Dispensers: This includes pharmacies - hospital, retail, and mail-order. They’re the last line of defense. If they can’t verify a drug, they can’t dispense it.

Real-World Impact: Successes and Failures
The results speak for themselves. CVS Health reported a 75% drop in suspect product investigations after automating their verification process. McKesson, one of the largest distributors, processed over 1.2 billion serialized transactions in 2023 with 99.98% accuracy. That’s nearly flawless. But not everyone got it right. In 2022, the FDA issued a warning letter to a regional distributor for failing to investigate and report suspect products - a direct violation of DSCSA. That’s the kind of failure the system was built to prevent. On the ground, pharmacists report mixed experiences. Some say scanning and verification now takes less than 30 seconds. Others still deal with delays caused by mismatched serial numbers or outdated software. Reddit threads from r/pharmacy in late 2023 were full of complaints: “My system says the barcode is valid, but the wholesaler’s data doesn’t match.” “We had to hold 400 boxes for three days because of a data glitch.” These aren’t just inconveniences. They’re risks. A two-day delay in verification means a potentially fake drug could sit on a shelf, waiting to be sold.Why This System Beats the Old Way
Before DSCSA, if a drug was recalled, pharmacies had to manually check paper records. If a batch was contaminated, they might have to pull every bottle of that drug - even if most were safe. Now, they can pinpoint the exact lot number and only remove the affected units. That saves money, reduces waste, and prevents unnecessary panic. The system also blocks diversion. That’s when drugs are stolen or illegally resold - often ending up on the black market. DSCSA’s traceability makes it nearly impossible to move stolen drugs without detection. According to McKesson, drug diversion dropped by 40% in the first five years of DSCSA implementation. Compared to the European Union’s Falsified Medicines Directive (FMD), DSCSA doesn’t require tamper-proof seals or a central government database. Instead, it relies on decentralized, interoperable data sharing. That’s simpler to implement, but it also means success depends entirely on every player following the same rules.What Happens After November 2024?
The November 27, 2024 deadline isn’t the end - it’s the starting line. The FDA will now shift from helping companies get set up to enforcing compliance. Companies that still can’t verify drugs will face fines, supply interruptions, or worse - being cut off from the legal supply chain. But the real story is what comes next. FDA Commissioner Dr. Robert Califf has said the agency is looking at expanding DSCSA to include certain over-the-counter (OTC) drugs - especially high-risk ones like insulin pens, painkillers, and asthma inhalers. Why? Because counterfeit OTC products are on the rise. A fake nicotine patch or fake Viagra can be just as dangerous as a fake prescription. Long-term, experts predict DSCSA will save $2.3 billion annually by 2027 through fewer recalls, less drug theft, and reduced healthcare costs from fake medications. PwC estimates counterfeit drug incidents will drop by 90% by then.
What You Can Do
As a patient, you don’t need to scan barcodes. But you can stay alert. If a pharmacy seems unsure about your medication - if they’re checking the bottle longer than usual, or if the packaging looks different - ask questions. Was this drug verified? Can you see the serial number? You can also ask your pharmacist if they’re DSCSA-compliant. Most are. But not all. If they’re not, they might be working with outdated systems that put you at risk. And if you ever see a drug sold online at a price that’s too good to be true - especially from a site that doesn’t require a prescription - don’t buy it. DSCSA protects the legal supply chain. It can’t stop illegal websites. That’s on you.Common Questions About DSCSA
What exactly does DSCSA track on each drug package?
Each package must have four key identifiers printed clearly: the National Drug Code (NDC), the lot number, the expiration date, and a unique 20-character serial number. These are encoded in both human-readable text and a machine-readable barcode. This combination allows pharmacies and distributors to verify the product’s origin and history electronically.
Does DSCSA apply to over-the-counter (OTC) drugs?
Currently, DSCSA only applies to prescription drugs. But the FDA has publicly stated it’s evaluating whether to extend the system to certain high-risk OTC products like insulin pens, asthma inhalers, and pain relievers. This expansion is likely within the next few years.
Why do some pharmacies still have delays in verifying drugs?
Delays happen when different software systems don’t communicate well. Even if two companies follow DSCSA rules, if one uses SAP and another uses TraceLink, data mismatches can occur - especially with serial numbers or lot codes. The FDA gave companies a year (2023-2024) to fix these issues. Most have, but smaller pharmacies still struggle with outdated tech or limited budgets.
Can counterfeit drugs still get through DSCSA?
It’s extremely difficult, but not impossible. DSCSA stops counterfeit drugs that enter the legal supply chain. But it doesn’t stop fake drugs sold online, smuggled across borders, or sold through illegal channels. That’s why it’s still important to buy medications only from licensed pharmacies - not websites or foreign vendors.
How much does DSCSA compliance cost a pharmacy?
For independent pharmacies, average implementation costs range from $150,000 to $200,000, covering new scanners, software upgrades, staff training, and system integration. Chain pharmacies spent millions - Walgreens invested $120 million between 2021 and 2022. But the long-term savings from fewer recalls and reduced theft far outweigh these costs.

Comments
Bridget Verwey March 6, 2026 AT 15:29
Okay but let’s be real - this system works like magic until your local pharmacy’s scanner starts acting up and they hold your insulin for 3 days because ‘data mismatch.’ I’ve seen it. My grandma’s blood pressure med got stuck in limbo. She was panicking. Meanwhile, the guy at the counter is scrolling through 17 error messages on a 2015 Dell. DSCSA sounds slick on paper. In practice? It’s a glorified IT nightmare with a FDA sticker on it. 😅
Andrew Poulin March 7, 2026 AT 19:05
This is why we need mandatory tech upgrades for small pharmacies. No more excuses. If you can’t scan a barcode in 2024 you shouldn’t be dispensing pills. Period.
Weston Potgieter March 9, 2026 AT 17:59
So basically the whole system is just a glorified barcode that everyone hates to use. Big pharma loves it because it makes them look responsible. Pharmacies? They’re drowning in compliance costs and half the systems still glitch. And don’t get me started on how many ‘verified’ packages turn out to be stolen from a warehouse in Ohio. This isn’t security. It’s theater with extra steps.
Vikas Verma March 10, 2026 AT 07:58
DSCSA implementation in US is a landmark achievement in pharmaceutical supply chain integrity. The interoperability via EPCIS standards enables real-time traceability which significantly reduces counterfeit infiltration. However, in emerging markets like India, adoption lags due to infrastructural constraints. Harmonization of global standards is imperative for true efficacy.
Sean Callahan March 11, 2026 AT 23:24
i just had to wait 45 mins for my zoloft because the system said the barcode was ‘unverified’ but the pharmacist swore it was legit… i just want my meds not a cybersecurity lecture. why is this so hard? also i think my pharmacy is using a flip phone as a scanner
phyllis bourassa March 13, 2026 AT 20:36
I love how we’re all acting like this is some revolutionary breakthrough. My local pharmacy still uses paper logs for 30% of their inventory. And the FDA’s ‘95% reduction’ in counterfeits? That’s based on reports they *could* verify - not what’s actually out there. You know what’s still floating around? Fake Cialis from Bangladesh sold on Instagram. DSCSA doesn’t touch that. It’s a closed-loop system for legal channels. Outside? Wild west.
William Minks March 15, 2026 AT 16:48
This is actually kind of amazing 🙌 I work in logistics and seeing how this system connects manufacturers to pharmacies across 50 states with zero central database? Mind blown. The fact that a mom in rural Nebraska can verify her child’s asthma inhaler in 12 seconds? That’s tech doing good. Kudos to everyone who made this work. 🏆
Jeff Mirisola March 16, 2026 AT 12:55
I’m not saying DSCSA is perfect - but it’s the first time in my 20 years as a pharmacist that I actually feel like we’re not just guessing if a drug is real. Before this? We’d cross-reference paper invoices and pray. Now? I scan, I verify, I move on. Yeah, there are glitches. Yeah, some systems are clunky. But the fact that we caught 12 fake batches last year alone? That’s lives saved. Don’t let the noise drown out the win.